ECG & Patient Monitoring Solutions for Global B2B Buyers
Professional supplier of ECG machines and patient monitoring equipment for distributors, hospitals, and healthcare projects. We support bulk purchasing, OEM medical device manufacturing, and long-term supply cooperation across global markets including South America, Southeast Asia, and the Middle East.
Certificates CE / FDA / ISO 13485
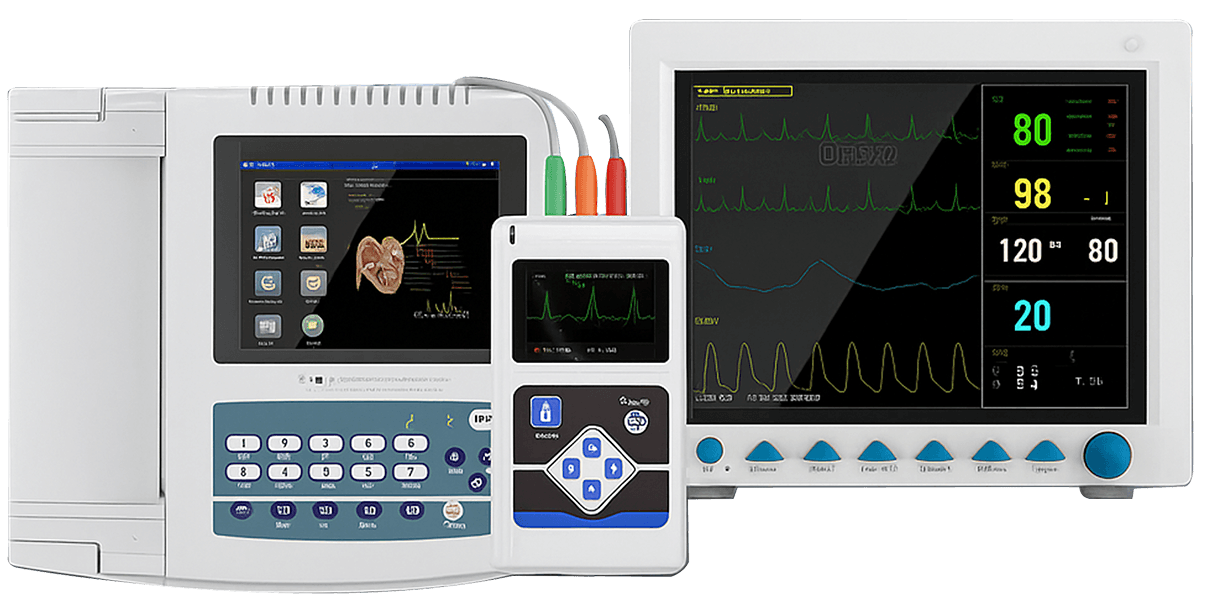

Why Choose Our Medical Device Supply
- Stable supply for bulk orders and long-term cooperation
- ECG and patient monitor products aligned with CE, ISO, and FDA requirements
- Experience supporting distributors and importers in multiple countries
- Documentation and compliance support for international market access
ECG Machines
Resting ECG systems designed for clinics, hospitals, and diagnostic centers.
Multiple channel options, reliable signal acquisition, and stable long-term performance.
Patient Monitors
Complementary Diagnostic Devices
Blood pressure monitors, pulse oximeters, and nebulizers to complete your product portfolio.
ECG Machines for Clinical & Distribution Use
Reliable ECG machines for hospitals, clinics, and distributors, including 3-channel, 6-channel, and 12-channel systems.
Patient Monitoring Solutions for Hospitals & Projects
Patient monitors designed for ICU, bedside, and portable applications, supporting continuous vital signs monitoring in hospitals and healthcare facilities.
Why We Focus on ECG & Patient Monitoring
“By focusing on these product categories, we help our partners build a consistent and scalable medical device portfolio instead of fragmented, short-term sourcing.”

Alex Hobbs,
Founder, CEO of Langhai
Supporting Medical Device Distribution in Global Markets
We provide ECG machines and patient monitoring devices for distributors and healthcare projects in regions such as Brazil, Mexico, Indonesia, and the Philippines.
Our solutions are designed to meet local regulatory requirements and market needs.
Stable Demand
Consistent market need across healthcare sectors
Scalable Supply
Bulk ordering capabilities for projects
Regulatory Clarity
Well-defined compliance pathways
Service Support
Long-term technical assistance
Solutions for Different Healthcare Needs
Our ECG and patient monitoring solutions cater to various healthcare settings and procurement requirements.
Solutions for Distributors, Hospitals & Healthcare Projects
Hospitals & Clinics

Distributors & Importers

Healthcare Projects & Tenders

OEM Medical Device Manufacturing & Private Label Support
We support OEM ECG machines and patient monitors for partners building their own brands.
Our focus is on long-term cooperation, stable product quality, and scalable supply.
Built for Professional Medical Procurement
Market Experience
Experience across multiple regional markets
International Compliance
Products aligned with major international compliance requirements
Documentation
Focus on documentation, communication, and supply stability
Bulk Supply
Designed for bulk supply and project-based cooperation
Products aligned with international medical standards, supporting CE, ISO, and FDA-related market requirements.
Insights & Buying Guides
- How to choose the right ECG machine for different markets
- Key considerations when sourcing patient monitors
- Basic certification knowledge for medical device importers
Contact a Reliable ECG & Patient Monitor Supplier
Tell us your market, application, or project requirements. We will recommend suitable ECG machines and patient monitoring solutions.
✔ Bulk supply ✔ OEM support ✔ Fast response
Address
Baoding City, Hebei Province, CHINA
Call Us
+86 173 3218 7072
Email Us
info@langhaimed.com
